Ammonia

Benzene
Acrolein

Nitric Acid
ILLUSTRATIONS
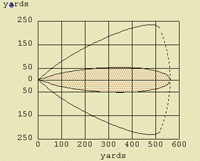
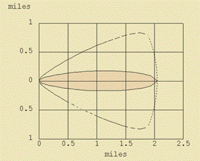
This section is designed to show examples of "footprints" (plume models) of certain chemicals under a variety of scenarios using ALOHA. The chemicals, ammonia, benzene, acrolein, and nitric acid, were chosen just to help illustrate certain concepts.
Ammonia produces a light gas, and benzene produces a heavy gas. They were chosen to show the differences between the "footprints" of spills of liquids that produce light and heavy gases.
 Ammonia |
 Benzene |
 Acrolein |
 Nitric Acid |
The charts and illustrations are meant to show some of the differences between the "footprints" of different chemicals. The amount released in each scenario is either
|
|

|
|
|

|
|
|

|
|
|

|
Ammonia 500 pounds |
 5,000 pounds |
 50,000 pounds |
 500,000 pounds |
These chemical spills are "modeled" at cool to hot temperatures, to illustrate the effect temperature has on dispersion of gases. The difference in daytime and nighttime models shows what effect the sun and warmth has.
DAY
|
NIGHT
|
Also, the models were conducted at 5 knots and 20 knots, to illustrate the difference between a small breeze and a brisk breeze on the dispersion of the gases.
Urban 5 k
|
Urban 20 k
|
When there is very little or no breeze (1.05 knott or less) ALOHA will usually show a circular "footprint." In these circumstances, ALOHA essentially predicts that there is a 1 out of 20 chance that the chemical spilled can be anywhere near the the site the chemical was spilled, even if the slight breeze is blowing in a different direction. This information is especially significant to responders during nighttime responses to spills because the chemical vapors could be moving in any direction, pooling, and not dissipating.

The difference in urban and rural (open country) are also illustrated.
Urban
|
Rural
|
One of the things illustrated here is that the "footprint" of a spill of a chemical does not correlate proportionately with the increase in the amount spilled. The "footprint" of a spill of a small tank of a chemical, 5,000 lbs, is not ten times the size of the "footprint" of a barrel, 500 pounds, of the same chemical. It varies, but a spill tens times the amount usually travels only two to three times the distance, with an increase in the overall area that could be affected by the spill. This is an important concept.
The "footprint" of a chemical may change in size and distance as more of the chemical is involved in the spill, but often the "footprint" remains essentially the same shape.
The effect that a strong breeze or a slow breeze has on the "footprint" is illustrated, as well as the difference between a rural setting with no trees or tall buildings and an urban setting with many obstacles to impede the spread of the plume of chemical vapors. The effect of temperature is also illustrated. A spill during cold conditions is different than in warm conditions.